H5N1, a highly pathogenic subtype of the avian influenza virus (bird flu), is not new. It has been with us since 1996, when it was first identified in domestic geese in Guangdong, China (CDC). H5N1 circulates primarily in animals (mostly birds) and can infect humans under specific exposure conditions. Diseases transmissible from animals to humans (or vice versa) are called zoonoses. By 1997, H5N1 had already killed six people in Hong Kong, and over the following two decades, H5N1 caused recurring waves across Asia, Africa and the Middle East, infecting almost one thousand people (WHO/PAHO).
As human hosts have no prior immunity against animal viruses, these tend to be highly virulent and lethal. Historically, one in every two people infected with H5N1 has died.
If zoonoses make the jump from animal to human, the high virulence combined with the capacity for respiratory spread and sustained human-to-human transmission increases the chances of a pandemic. These are the so-called viruses with pandemic potential. In addition, H5N1 as well as other influenza strains, are RNA viruses with a segmented genome consisting of eight separate fragments. When two different influenza strains simultaneously infect the same cell, those segments mix freely and can reassemble into new combinations. This process is called reassortment or antigenic shift. The result is a virus with radically different properties. Influenza viruses also undergo antigenic drift, which is the gradual accumulation of small mutations that occurs in all RNA viruses over time. Influenza does both: it drifts constantly, and occasionally shifts. This is why influenza viruses, which combine these recombination mechanisms with respiratory transmission, have driven major pandemics throughout history: 1918 (Spanish flu, 50–100 million deaths), 1957 (Asian flu), 1968 (Hong Kong flu), and 2009 (swine flu, H1N1), which originated through reassortment in pigs in Mexico.
In 2020, a new variant of H5N1 emerged in wild ducks, first detected in the Netherlands: clade 2.3.4.4b (Frontiers in Veterinary Science). Although its parent lineage had been circulating in Asia since 2013-2014, this was the first time it established itself permanently in wild aquatic bird populations across Europe and Asia, shifting from seasonal to year-round circulation. Unlike previous H5N1 variants, which disappeared from wild bird populations each summer and had to be reintroduced each winter by migratory birds arriving from Asian reservoirs, clade 2.3.4.4b no longer depended on seasonal migration to survive. It was always there, in the ducks, gulls and sandpipers that cross continents.
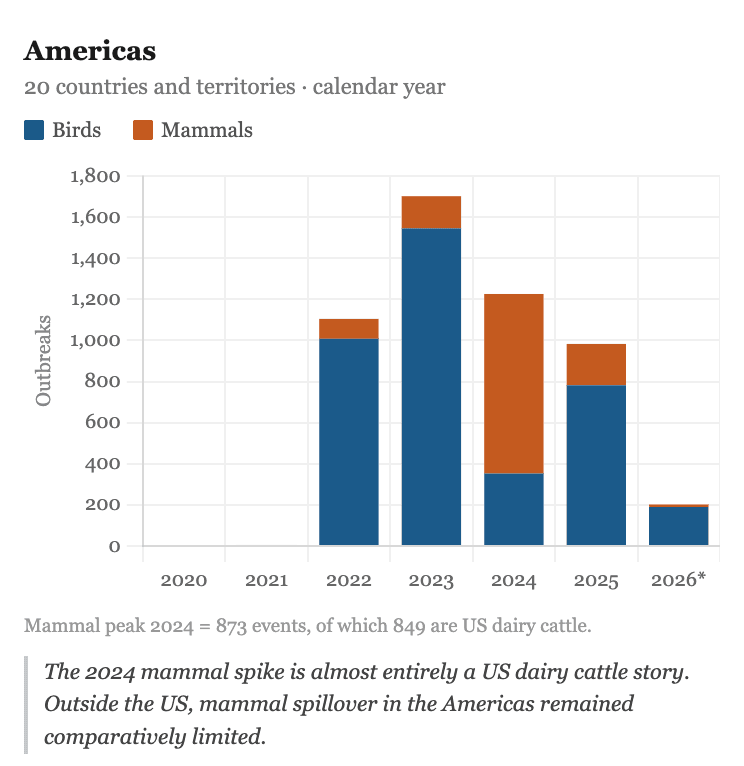
It then crossed the Atlantic, reaching Canada in autumn 2021, and spread to Central America and South America in 2022. Between 2022 and 2023, massive die-offs of marine mammals (sea lions, elephant seals) and birds (penguins) began appearing along the Pacific and Atlantic coasts of South America. Thousands, then tens of thousands of carcasses on the beaches of Peru, Chile and Argentina. At Península Valdés, 97% of elephant seal pups born that season died (Uhart et al., Nature Communications, 2024). These deaths implied that the virus was not producing isolated spillover cases, jumping from birds, its natural reservoir, to marine mammals, but was already transmitting from mammal to mammal, something H5N1 had never demonstrated at this scale in wildlife.

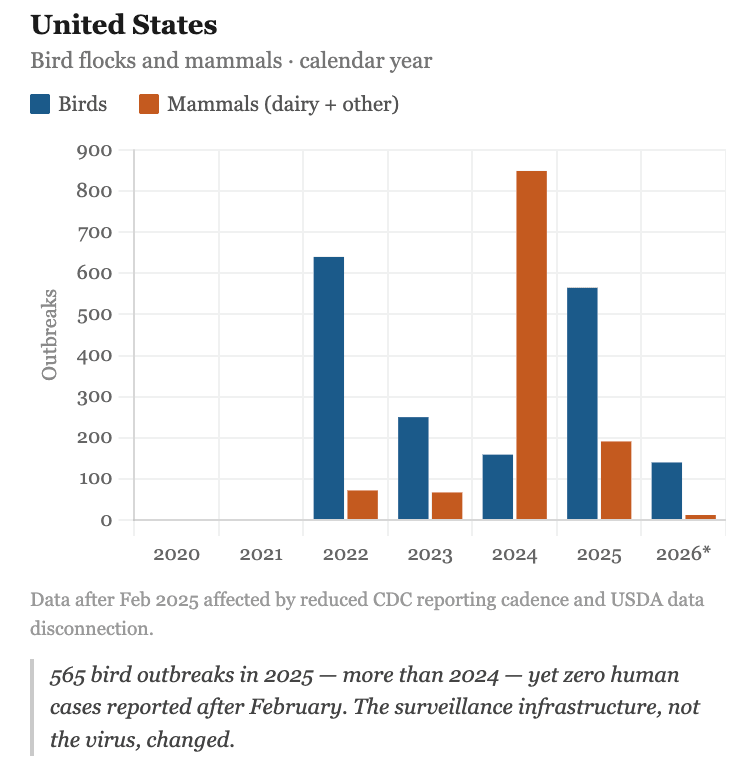
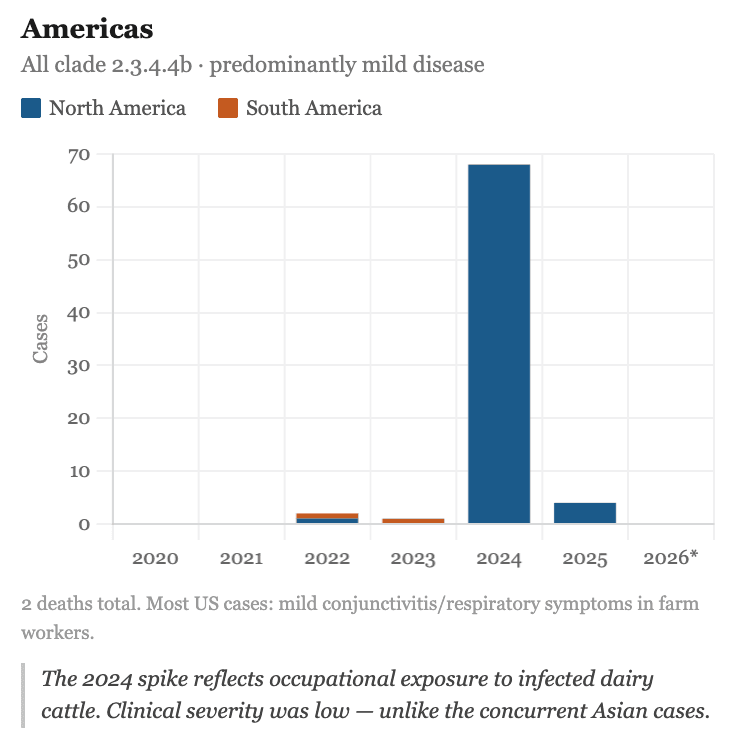
In March 2024, it appeared in dairy cattle in Texas (USDA APHIS), spreading to more than 849 farms across 17 US states. The virus was already stably circulating in US dairy cattle, with the capacity for transmission through raw milk and aerosol generated during milking. It was also infecting domestic cats. In 2024, the first human cases began to be reported: 67 people infected that year, the majority with mild infections, all farm workers with direct exposure to infected animals (CDC).
In January 2025, at the peak of the epidemic, the system responsible for monitoring all of this began to be dismantled.
Epidemiological censorship with consequences yet to be seen
The new US administration ordered a pause on all external communications from the CDC (CNN, January 2025). The Morbidity and Mortality Weekly Report (MMWR), published without interruption since 1952, missed two consecutive issues for the first time in its history due to the HHS communications freeze, and resumed publishing with politically directed content. Studies on transmission of the virus to domestic cats owned by dairy workers were blocked (CBS News, February 2025). In July, the CDC reduced its H5N1 reporting from weekly to monthly and stopped publishing USDA animal data from its website (CDC, July 2025). The US also withdrew from the WHO in January 2025 (effective from 2026), withdrawing its entire contribution, 18% of WHO’s total funding and ceased negotiations on the WHO Pandemic Agreement, and rejected the amended International Health Regulations (White House, January 2025), the legal framework governing global cooperation on disease control across borders. As US H5N1 reporting went silent – no human cases have been reported since February 2025 – international coverage also declined, despite the epidemiological situation has continued to escalate.
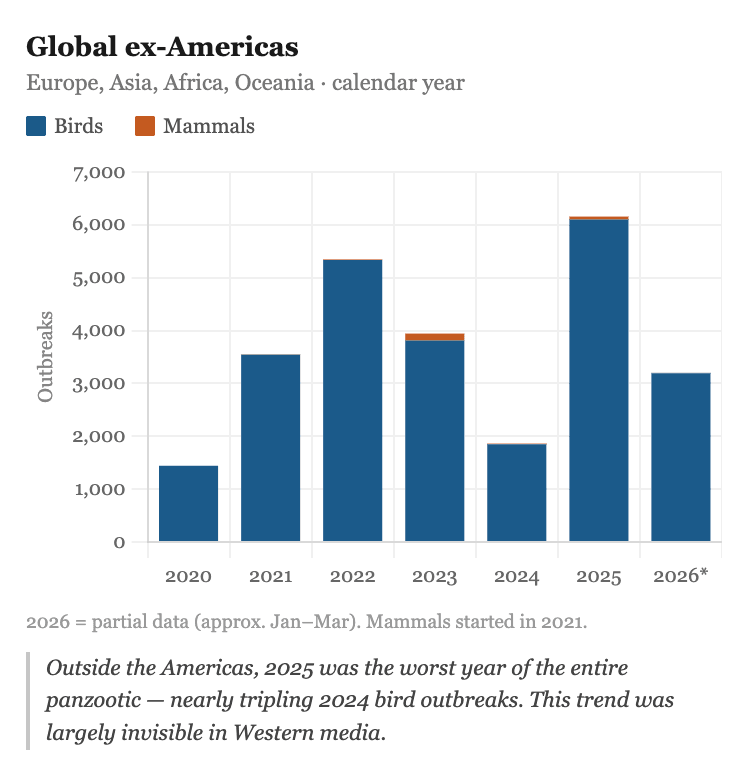
Data from the FAO, the WHO and national authorities that have maintained epidemiological surveillance show that 2025 was the worst year of the entire clade 2.3.4.4b era, and the trend for 2026 continues to rise (FAO H5 HPAI Dashboard).The situation has continued to escalate through 2026. In February, H5N1 was confirmed for the first time in northern elephant seals in California, the first detection in that species in North America, and a reminder that the virus continues expanding into new mammalian hosts. In March 2026, EFSA documented the first possible spillover to dairy cattle in the European Union (EFSA, March 2026). Note: 2026 data in the charts below is incomplete as it covers only the first three months of the year, which explains the apparent decline.
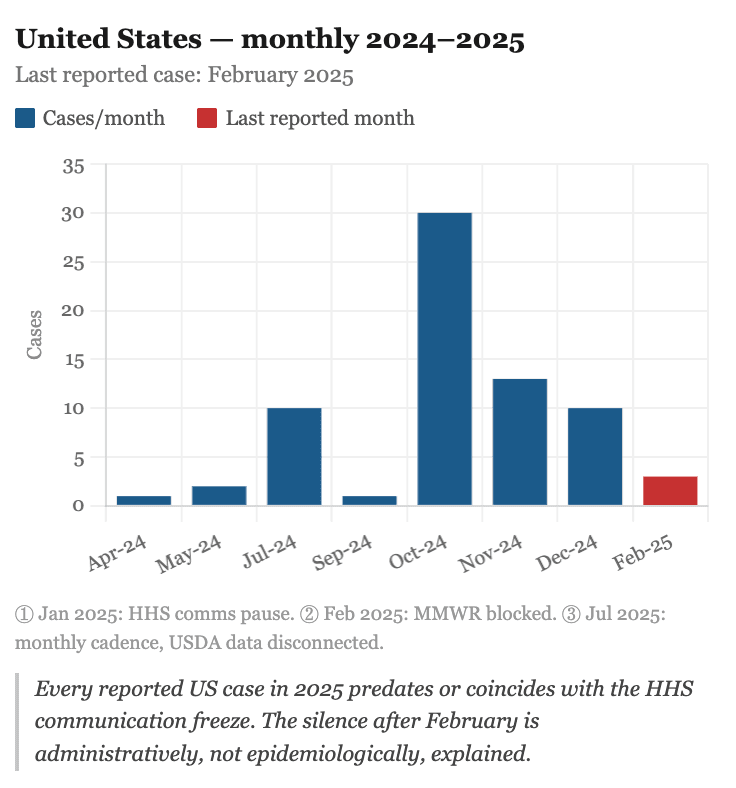
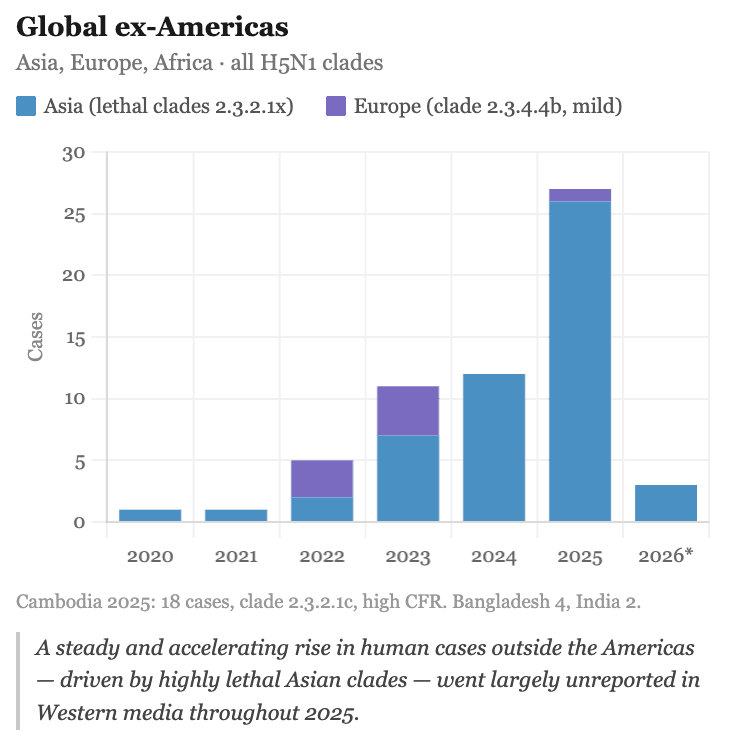
The H5N1 virus has not disappeared. It has broken records while the world looks the other way.
Highly Pathogenic Avian influenza is not COVID. There is no sustained human-to-human transmission yet. But now is precisely when vigilance must be maintained, not after the virus mutates into a form transmissible between humans, triggering a pandemic. Public health surveillance systems serve to detect these changes before they become emergencies, enabling us to prepare for both prevention and response. Dismantling and censoring these surveillance systems during an active outbreak, and dragging the world’s media agenda along with that decision, could have potentially catastrophic consequences.
Sources
| H5 HPAI animal outbreaks by country and year | FAO Avian Influenza Dashboard |
| Human H5N1 cases by region and month | Our World in Data / WHO Global Influenza Programme |
| Situation in the Americas | PAHO/WHO, March 2026 update |
| Global HPAI situation reports | WOAH HPAI Situation Reports |
| US animal detections | USDA APHIS |
| US human surveillance | CDC Bird Flu |
| Mammal-to-mammal transmission in pinnipeds | Uhart et al., Nature Communications, 2024 |
| Global panzootic in mammals | Peacock et al., Nature, 2025 |
| Surveillance gaps in mammals | OFFLU, March 2026 |
| CDC interference and MMWR censorship | CBS News, February 2025 |
| Impact of cuts on outbreak response | NBC News, February 2025 |
| Clade 2.3.4.4b emergence and spread | Frontiers in Veterinary Science, 2024 |
© Episphera, 2026. Licensed under CC BY 4.0.
